Division of Drug Information
Center for Drug Evaluation and Research Food and Drug Administration
10903 New Hampshire Ave.
Building 51, Room 2201
Silver Spring, MD 20993
Submitted by email September 16, 2014
RE: Docket No. FDA-2014-D-0397-0001
RE: Draft Guidance for Industry on Internet/Social Media Platforms With Character Space Limitations: Presenting Risk and Benefit Information for Prescription Drugs and Medical Devices; Availability
The FDA released draft guidance in June 2014 regarding the use of platforms with character space limitations, calling for comments. The Digital Health Coalition (DHC) congratulates the FDA on its release of this additional draft guidance and the invitation to comment. We believe it is a strong step forward towards our collective goal of using digital innovation to improve patient health outcomes. The Digital Health Coalition, a nonprofit organization with 501(c)(3) status, was created to serve as the collective public voice and national public forum for the discussion of the current and future issues relevant to digital and electronic marketing of healthcare products and services. We engage multiple and diverse stakeholders–including pharmaceutical and biotech manufacturers, physician and patient groups, advertising agencies, health information providers, internet search companies, and medical technology companies. We are informed by a diverse advisory board as well as approximately 100 “DHC Health Scholars” with representation from all industry sectors.
The DHC submits these comments to identify a few elements of this draft guidance that we strongly encourage FDA to review. It is our belief that with the wide consumer adoption of the platforms under discussion, there exists a significant opportunity to connect with patients, and partner with them during the healthcare journey. It remains the DHC’s view that any FDA guidance should have the positive effect of encouraging pharmaceutical companies to increase their level of participation in social media and promoting the flow of information, ultimately for the benefit of patients and healthcare in general.
The DHC’s core value is a belief that regulated healthcare companies should endeavor to participate in social media as a means to promote public health, improve patient outcomes and facilitate productive patient/physician relationships (DHC Social Guiding Principles, 2012). Our comments here are designed to help bring this draft guidance to a place that fosters and encourages meaningful and impactful participation. The social media landscape is varied and each platform displays content in different ways; the DHC urges the FDA to take into account the differentiation of platforms and to allow flexibility in how each platform showcases the necessary information to consumers. First, we would like to address the Twitter examples used in the draft guidance. As a part of the process for assembling these comments, we conducted a survey of industry members that we designed to help us better understand the current importance and perceived potential of the types of platforms under discussion, including Twitter. We believe the results of that study could help frame the comments included for FDA. A summary of the findings is also being provided to FDA.
Highlights from the survey that we believe would be of interest to FDA include:
Is it important to your business to communicate to your primary target audience on Twitter?
| Response | Count | Percent | |||
| Yes | 12 | 75.0% | |||
| No | 4 | 25.0% | |||
| Who is your primary target audience on Twitter? | |||||
| Response | Count | Percent | |||
| Patients | 7 | 87.5% | |||
| Healthcare professionals | 1 | 12.5% | |||
| Caregivers | 0 | 0.0% | |||
| Do you agree or disagree with the following statements? | ||||
| AGREE% | # | DISAGREE% | # | |
| The draft guidance on platforms with character space limitations led to increased discourse within my team/company over the use of Twitter | 75.0% | 12 | 25.0% | 4 |
| There is an opportunity to positively impact patient health outcomes through the use of Twitter | 81.3% | 13 | 18.8% | 3 |
| The draft guidance leaves open the door to communicate with patients via Twitter | 75.0% | 12 | 25.0% | 4 |
| The draft guidance leaves open the door to communicate with health professionals via Twitter | 68.8% | 11 | 31.3% | 5 |
| My company intends to respond to the platforms with character space limitations FDA draft guidance by submitting comments (to FDA) | 66.7% | 10 | 33.3% | 5 |
| I believe Twitter has the potential to facilitate customer service between patients and pharmaceutical/device companies | 100.0% | 16 | 0.0% | 0 |
| I believe Twitter has potential for branded advertising campaigns by pharmaceutical/device brands trying to reach patients | 68.8% | 11 | 31.3% | 5 |
| I believe Twitter has potential for branded advertising campaigns by pharmaceutical/device brands trying to reach health professionals | 75.0% | 12 | 25.0% | 4 |
On Fair Balance In Twitter-Type Platforms
***********************
FDA Draft Guidance:
Risk information should be presented together with benefit information within each individual character-space-limited communication (e.g., each individual message or tweet).
The content of risk information presented within each individual character-space-limited communication should, at a minimum, include the most serious risks associated with the product.
***********************
Considering only text character allowance and not the rich media options that accompany text ignores some of the strongest elements of the patient communication opportunity. In the absence of clearly defined parameters, the industry common practice is to avoid the opportunity entirely. It is the opinion of the DHC that this would be a loss for the patient and caregiver.
Given the widespread public adoption, platforms with character space limitations, such as Twitter, represent what we believe to be a great opportunity to communicate with patients. However, without clarifying guidance that factors in the full landscape possibilities, it’s likely that prescription drug and medical device companies will only consider the specific examples in the current draft guidance. For example, when reviewing the current draft guidance and provided Example 1c,
NoFocus (rememberine HCl) for mild to moderate memory loss-May cause seizures in patients with a seizure disorder www.nofocus.com/risk,
our industry survey revealed a strong consensus that, while accurately depicting one method of communicating via Twitter, the example does not capture the complete breadth of Twitter communication and would not be effective in impacting patient health outcomes. In other words, the current draft guidance would heavily discourage “branded tweets,” which, if utilized, can provide effective information to patients in an easily accessible and mainstream way. We urge the FDA to consider more examples of ways that social platforms, such as Twitter, could be successfully used, and then revise the draft guidance to best capture that potential, for the sake of the patient.
It is important to consider that a tweet contains more information than just 140 characters. For example, a feature known as “Twitter Cards” (more information can be found at https://dev.twitter.com/docs/cards) allows for rich media to supplement text-based Tweets with additional content and context.
This additional Tweet context can be an image, video, or featured website content. Such rich media would allow advertisers to include necessary disclosures outside of the character restraints of a Tweet.
Examples of Twitter Cards include:
Website card: Features a clickable image and a button that allows users to be directed to a website URL with one click from the Tweet
Video card: Allows videos to be played alongside a Tweet and within the user’s timeline without being redirected to another site
Photo card: Displays a selected image directly in a Tweet
Using the hypothetical provided in the “Internet / Social Media Platforms with Character Space Limitations” June 2014 Draft Guidance, NoFocus, a physical representation of the Draft Guidance’s Example 1c tweet would only include text and a URL link (see Image 1).
Image 1:
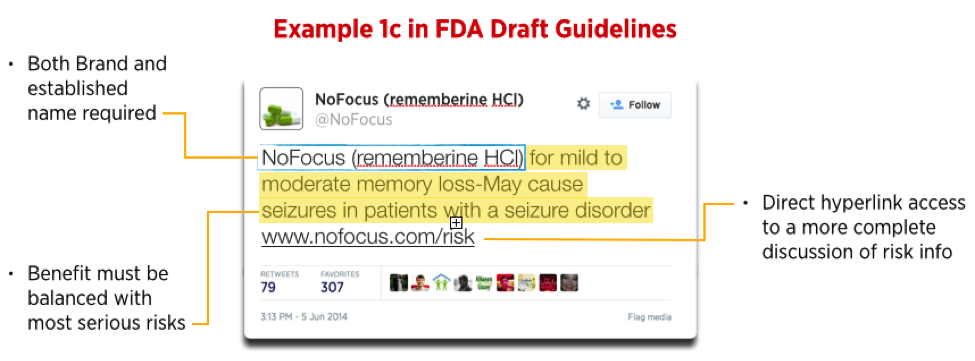
By using rich media options (such as Twitter Cards), however, prescription drug and medical device manufacturers have the ability to supplement a tweet with an image, video, or featured website content. In order to address fair balance and to direct users to the Important Safety Information (ISI) from a URL link, a Twitter Website Card, for example, could include an image with additional text to cover fair balance and a prominently displayed URL link that direct users directly to the ISI. Image 2 shows how this may be accomplished using the NoFocus hypothetical used in the draft guidance.
Image 2:
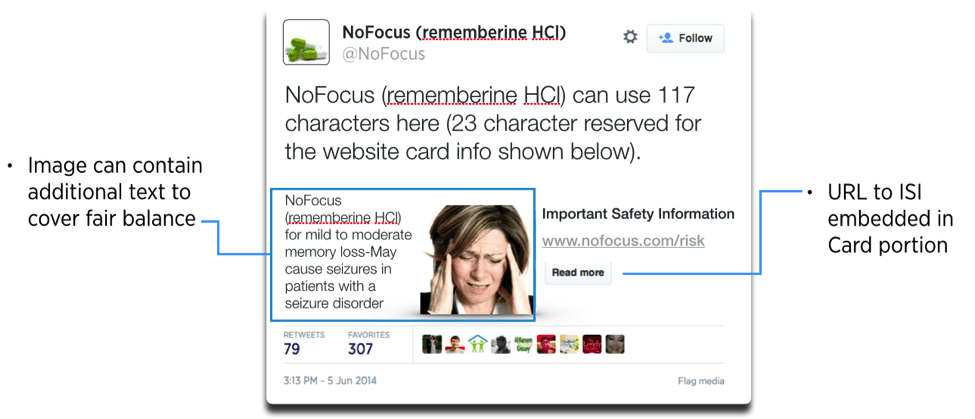
A comparison of Image 1 and Image 2 highlights that through the use of Twitter Cards, additional necessary information can be added to a tweet. This example using a Twitter Website Card (Image 2) is just one example of rich media that can be included with a tweet in order to present important information to patients.
The DHC urges the FDA to review the draft guidance with consideration for the ways branded communication on platforms with character space limitations could have a positive impact on patient health outcomes, especially in taking a deeper look at rich media options.
In the provided example, Twitter rich media options provide a similar tableau as print advertisements, which already provide adequate product claim ads to patients. In print ads, the included “fair balance” text is provided in proximity to the product claim ad but is found on a facing or reverse page. The consumer must still take action to see the full fair balance which is not always occupying the same visual space. We believe applying this proven approach to platforms with character space limitations that provide rich media options would allow for more robust communication with patients while still meeting the fair balance requirement.
Questions for FDA:
● There is some confusion around the stipulation within this draft guidance that there be a dedicated (and named) link to risk information. It was believed, based on prior statements by FDA, that linking to a PI/ISI on a landing page was sufficient. Is that no longer the case?
● If, staying with the provided example, the Twitter account was “@NoFocus”, it is assumed that the name would not need to be repeated in the tweet text, but we would like clarification.
● While the provided example demonstrated a product label that fit (albeit eating up 12 of 140 characters) what is the opportunity “cut-off” when the product label is too complex to deal with the 140 character constraint? Are further abbreviations of product/label acceptable in Tweets?
Although the current rich media options on Twitter enable for more information to be displayed in a tweet, it is also important to issue a final guidance that would provide broad guidance, ensuring that all technology platforms have the ability to innovate and to evolve their respective platforms towards improving patient communication. New methods of displaying information will surely come over time and it would be prudent to allow for those changes as long as the consumer is able to easily access both the information and risks associated with any pharmaceutical product.
On Use of Google sitelinks
In addition, the DHC would like to encourage the FDA to revisit the recommendations regarding how to achieve fair balance where Google sitelinks are concerned. It is the understanding of the DHC that the proposed use of Google sitelinks in order to achieve fair balance is not viable. Based on the current industry understanding of the Google sitelinks platform, there appear to be several factors that would ultimately determine whether or not the sitelinks would appear, all of which are beyond the control of the pharmaceutical or medical device company who might place the ad. Unless a manufacturer can confidently run the ad as designed, it would be a mistake to attempt to use sitelinks for the purpose of fair balance, and we are sure FDA will agree upon review.
Once again, the DHC commends the FDA on continuing to wrestle with the changing landscape created by digital innovation and ever-evolving social media. We thank the FDA for the opportunity to comment on the draft guidance and offer the Digital Health Coalition’s assistance in helping to resolve, provide supporting information or further clarify the questions its members have presented as part of our comments.
Kindest Regards,
Christine Franklin
Managing Director
Digital Health Coalition
digitalhealthcoalition.org
244 5th Avenue
NYC NY 10001
(800) 261-1655
